












Simplifie et parfait le traitement implantaire grâce à un protocole digital révolutionnaire, unique, et breveté.

Des caractéristiques éprouvées depuis 30 ans et validées par des organismes indépendants.

Une sélection de 3 marques de scanners intra-oraux performants pour tous les budgets.
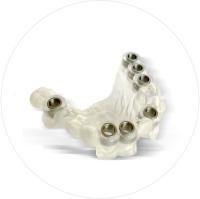
Une trousse simple et un système ergonomique apportant confort de travail et sécurité.


La Dental App regroupe tous nos services pour vous simplifier la compréhension, et l’usage de nos produits. Elle vous permet d’échanger avec LYRA ETK comme un partenaire de production sur mesure, et d’avoir accès à toute heure à l’ensemble des travaux en cours. Un service full digital pour des informations et des réalisations rapides et précises.









LYRA ETK est une Medtech française à vocation mondiale spécialisée dans la restauration dentaire.
Les innovations de LYRA ETK ont pour objectif de proposer de nouveaux traitements et protocoles pour mieux soigner vos patients, et répondre à leurs attentes en terme d’esthétique, de rapidité, de fonctionnalité et de confort. Leurs performances sont validées et reproductibles grâce à des protocoles codifiés très précis s’appuyant sur la science.
La proximité est inscrite dans l’ADN de LYRA ETK. Nous cultivons une relation étroite avec les chirurgiens-dentistes et laboratoires qui ont contribué à bâtir notre expertise et notre compréhension des contraintes du cabinet et du laboratoire. Nous sommes engagés dans l’accompagnement des praticiens et prothésistes pour développer leur activité.
Nos produits sont assortis de protocoles garantissant la prévisibilité et la reproductibilité des résultats. Ces protocoles sont codifiés pour guider au maximum l’équipe de soin.
L’équipe LYRA ETK est agile et réactive. Elle s’inspire de la veille technologique et de vos besoins pour faire évoluer constamment ses gammes.
Nous nous engageons à contribuer à la réduction des déchets et à la protection de l’environnement en retravaillant nos packagings, en traitant ou en recyclant nos déchets, en limitant notre consommation d’énergie.
Formation et accompagnement des praticiens
